Vaccines need specific types of storage to remain safe.
 Storage of vaccines is important — so important that when* the New York State’s Vaccine for Children (VFC) Program1 informed all enrolled providers that they now had to move to stand-alone units, they included four pages of specifics about freezers and refrigerators. If you look at the Centers for Disease Control and Prevention (CDC) Vaccine Storage and Handling Tool Kit2, it dedicates eight pages to Vaccine Storage and Temperature Monitoring Equipment, not including Emergency Storage and Handling guidance. Traditionally, vaccines have been stored in Refrigerators (+2° to +8°C) or Freezers at –20° C but with some of the early COVID-19 vaccine candidates, ultralow storage (–80°C) will be added to the “vaccine storage” mix. The goal of mitigating the risk of vaccine loss due to the accidental freezing or warming associated with storage is becoming more complicated than ever.
Storage of vaccines is important — so important that when* the New York State’s Vaccine for Children (VFC) Program1 informed all enrolled providers that they now had to move to stand-alone units, they included four pages of specifics about freezers and refrigerators. If you look at the Centers for Disease Control and Prevention (CDC) Vaccine Storage and Handling Tool Kit2, it dedicates eight pages to Vaccine Storage and Temperature Monitoring Equipment, not including Emergency Storage and Handling guidance. Traditionally, vaccines have been stored in Refrigerators (+2° to +8°C) or Freezers at –20° C but with some of the early COVID-19 vaccine candidates, ultralow storage (–80°C) will be added to the “vaccine storage” mix. The goal of mitigating the risk of vaccine loss due to the accidental freezing or warming associated with storage is becoming more complicated than ever.
Working together, the World Health Organization (WHO) and jurisdictions at many levels develop guidance, and it’s critical to stay connected with the appropriate manufacturers and government agencies to ensure compliance and to develop best practices for vaccine storage. In the case of managing vaccines at ultralow temperatures, we are still waiting for the CDC to update the Vaccine Storage and Handling Tool Kit but regarding storing vaccines in refrigerators and freezers there is a large amount of experience that has been established.
6 factors and best practices to consider when selecting refrigerators and freezers for vaccine storage:
1. Use purpose-built, pharmaceutical-grade or household-grade units designed to either refrigerate or freeze — Do Not Use dormitory-style or bar-style combined refrigerator/freezer style units under any circumstances.
2. Consider using safeguards to ensure the door of your storage unit stays closed such as door alarms or door locks.
3. All vaccine storage units require either a built-in microprocessor with a digital sensor or a stand-alone digital data loggers with a buffered temperature probe.
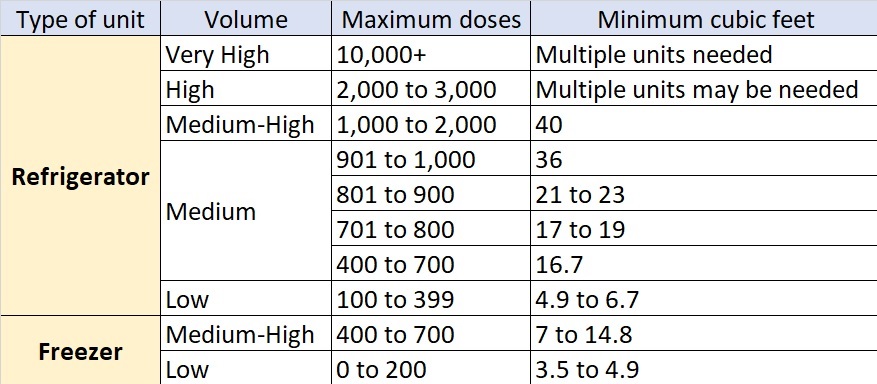
4. Vaccine storage units need to offer enough space to accommodate your maximum vaccine inventory without crowding. The recent New York State program update offered guidance in this respect. See the table below:
5. Reduce the possibility of power interruption to the unit:
- Plug in only one storage unit per electrical outlet
- Label fuses and circuit breakers to not turn off power
- Reduce the chances of accidentally unplugging the storage units with safety locks and warning signs
6. Place vaccine storage units in a well-ventilated area away from heat sources including direct sunlight.
There are many factors and best practices that go into mitigating the risk of vaccine loss due to the accidental freezing or warming, but at the core is a vaccine storage unit that will maintain its temperature.
Cole-Parmer offers a wide range of refrigerators, freezers and ultralow units that address this core confidence. Let us know how we can help.
References:
- Center for Disease Control and Prevention, CDC Vaccine Storage and Handling Kit, Accessed, October 15, 2020. https://www.cdc.gov/vaccines/hcp/admin/storage/toolkit/storage-handling-toolkit.pdf
- State of New York, New York State Vaccines for Children Program (NYS VFC) Vaccine
Stand-Alone Storage Unit Purchasing Guidance, Accessed October 14, 2020. www.health.ny.gov/prevention/immunization/vaccines_for_children/docs/storage_unit_purchasing_guidance
Learn more about the vaccine cold chain: The Cold Chain Facilitating Vaccine Distribution



Be the first to comment on "6 Factors to Consider When Selecting Refrigerators and Freezers to Store Vaccines"